We are helping bring customer-centricity into MedTech business strategies
Strategic research shaping customer-centric MedTech growth
New Product Research
Human-Centered Insight for Meaningful MedTech Innovation

Clinical workflows are evolving fast. Healthcare systems demand efficiency. Medical device innovation can no longer happen in isolation. Successful new product introductions require a deep understanding of real-world clinical environments, human behavior, and unmet needs that traditional research often misses.
At EMeRG, we believe breakthrough medtech innovation starts with human-centered insight. Our New Product Introduction (NPI) research helps medical device companies move beyond assumptions. We support the design of solutions that are clinically relevant, adoptable, and scalable. Our work has enabled the launch of medtech products valued at over $1.1B globally.
Insight Beyond the Obvious
Traditional research captures what customers can say. EMeRG’s ethnography-led, contextual research goes deeper. Through Voice of Customer (VoC) research, clinical immersions, and ethnographic studies, we uncover what truly drives usage and adoption.
Our research reveals:
- Unarticulated and latent unmet needs
- Rational and emotional drivers of adoption
- Clinical, ergonomic, and workflow requirements
- Hidden motivators and barriers to trial and use
- Everyday pain points and inefficiencies
- Clear input on new concepts, prototypes, and value propositions
- Real-world brand perception
From Insight to Innovation
We convert insight into clear direction. Our narratives align teams and guide decisions. By grounding innovation in lived clinical experience, EMeRG helps medtech organizations reduce risk, accelerate adoption, and deliver products that improve care.

How We Have Helped Med-tech Firms in New Product Introductions and Development:

Outcomes Research Capabilities
Evidence That Drives Clinical Adoption, Market Access, and Commercial Value
Clinical expectations are intensifying. Health systems increasingly demand demonstrable clinical value, operational efficiency, and financial sustainability before adopting new technologies. Innovation alone is no longer sufficient. Adoption today depends on robust, real-world evidence that clearly translates performance into measurable outcomes.
At EMeRG, outcomes research is positioned as a strategic adoption enabler rather than a post-hoc validation exercise. Over the past decade, we have delivered 40+ outcomes and evidence generation studies across 25+ countries, supporting medical technology programs spanning clinical validation, economic value demonstration, and adoption strategy. Our work has contributed to commercialization pathways for medtech innovations collectively exceeding USD 1B in global market value, helping organizations strengthen market access, accelerate adoption, and sustain competitive differentiation.
Wish to know more about our Outcomes Research services?

Real-World Outcomes and Observational Studies
Demonstrating technology performance in routine clinical practice
Study approach
- Pre-post implementation outcome assessments
- Prospective and retrospective observational studies
- Workflow and time-motion analysis
- Comparative real-world performance evaluations
Business value created
- Strengthens clinical credibility and adoption confidence
- Supports procurement, tender, and reimbursement discussions
- Converts clinical performance into commercially relevant evidence
Workflow, Usability, and Adoption Research
Optimizing technology integration into clinical environments
Study approach
- Ethnographic workflow observation and human factors evaluation
- Structured usability testing and adoption barrier analysis
- Productivity and throughput impact assessments
- User experience and clinician perception studies
Business value created
- Improves technology adoption and utilization rates
- Supports operational ROI positioning for providers
- Informs deployment, training, and customer success strategies
Access, Equity, and Care Delivery Impact Studies
Understanding technology reach and health system impact
Study approach
- Assessment of technology deployment across urban, rural, and resource-constrained settings
- Analysis of care pathway redesign enabled by technology adoption
- Evaluation of service availability, referral patterns, and wait-time reduction
- Utilization and reach analysis across population segments and geographies
Business value created
- Enables expansion into underserved and emerging markets
- Strengthens policy and public health engagement
- Positions technology as a care access enabler
Health Economic and Value Evidence Studies
Translating clinical outcomes into financial impact
Study approach
- Cost-effectiveness and cost-utility analyses
- Budget impact modelling and financial outcome studies
- Total cost of ownership and lifecycle cost evaluation
- Economic evidence synthesis for market access
- Meta-analysis and indirect comparative modelling
- Health economic simulation and decision analytic models
- Epidemiology, utilization, and adoption forecasting
Business value created
- Supports reimbursement, pricing, and market access strategy
- Strengthens competitive positioning with quantitative evidence
- Demonstrates financial value to providers and payers
- Strengthens value-based selling narratives
Evidence Synthesis and Literature Intelligence
Structuring existing evidence for strategic positioning
Study approach
- Systematic and targeted literature reviews
- Evidence gap and competitive benchmarking analysis
- Scientific landscape assessments
- Structured clinical evidence synthesis
Business value created
- Identifies differentiation opportunities and evidence gaps
- Supports scientific differentiation and regulatory positioning
- Strengthens clinical messaging and regulatory submissions
- Guides future evidence generation priorities
GTM Strategy Research
Designing Scalable Market Access for MedTech
In complex and fragmented healthcare markets, product success depends on more than innovation. It requires a clear, execution-ready go-to-market strategy. This is especially true in emerging markets, where channel dependence, infrastructure gaps, and local competition shape outcomes.
At EMeRG, we help medical device companies design market-rooted GTM strategies that balance speed, control, and scale. Our research-led approach moves beyond generic models to reflect real customer behavior, competitive dynamics, and on-the-ground realities.
GTM Insight for Emerging Markets
In regions such as ASEAN and Africa, limited direct presence and reliance on channel partners make GTM decisions critical. Success requires a deep understanding of customer segments, care delivery infrastructure, and local competitive forces. EMeRG supports clients in identifying optimal routes to market and building differentiation at the point of care.

How we have helped med-tech firms with go-to-market research in emerging markets:
Our GTM Research Focus
We provide clarity across the commercialization ecosystem, including:

Market challenge identification and validation

Needs-based customer and stakeholder segmentation

Competitive landscape assessment

Distribution strategy and channel partner due diligence

Value proposition, messaging, and pricing assessment
Sales, service, and customer engagement models
From Strategy to Execution
We translate insight into clear strategic choices and practical GTM playbooks. By grounding strategy in local market realities, EMeRG helps medtech companies reduce risk, accelerate adoption, and scale with confidence.
GTM Strategy Research
In several emerging markets where medical device sales are predominantly dependent on channel partners, formulating the right go-to-market strategy requires profound understand of the market dynamics.
EMeRG continues to work with medical device companies to evaluate the optimal approach to reach end customers with distinct competitive advantages. In ASEAN and African regions where most medical device companies struggle with setting up direct presence, understanding distribution options requires in-depth understanding of customer segments, infrastructure and local competitive landscape. The key cornerstones of our GTM research includes:

Market challenge identification and validation

Needs-based customer segmentation

Competitive landscape evaluation

Distribution assessment including channel partner due diligence

Value proposition and messaging testing

Pricing assessment

Sales and services inclusions
Customer engagement and satisfaction tracking
Pricing Research
Translating Clinical Value into Sustainable Revenue
In today’s medtech landscape, pricing is no longer just a commercial decision. It is a strategic signal of value, differentiation, and long-term viability. As medical device companies navigate the balance between value-driven and premium offerings, defining the right pricing strategy has become both complex and critical.
At EMeRG, we help medtech organizations move beyond cost-plus or competitor-led pricing. Our pricing research is designed to uncover how different stakeholders—clinicians, procurement teams, administrators, and payers—perceive, prioritize, and trade off value in real-world decision-making.
Pricing Insight Beyond Stated Preferences
Customers often struggle to articulate the value of individual features in isolation. EMeRG’s advanced pricing methodologies are built to reveal true willingness to pay, trade-offs, and price sensitivity within realistic choice contexts.
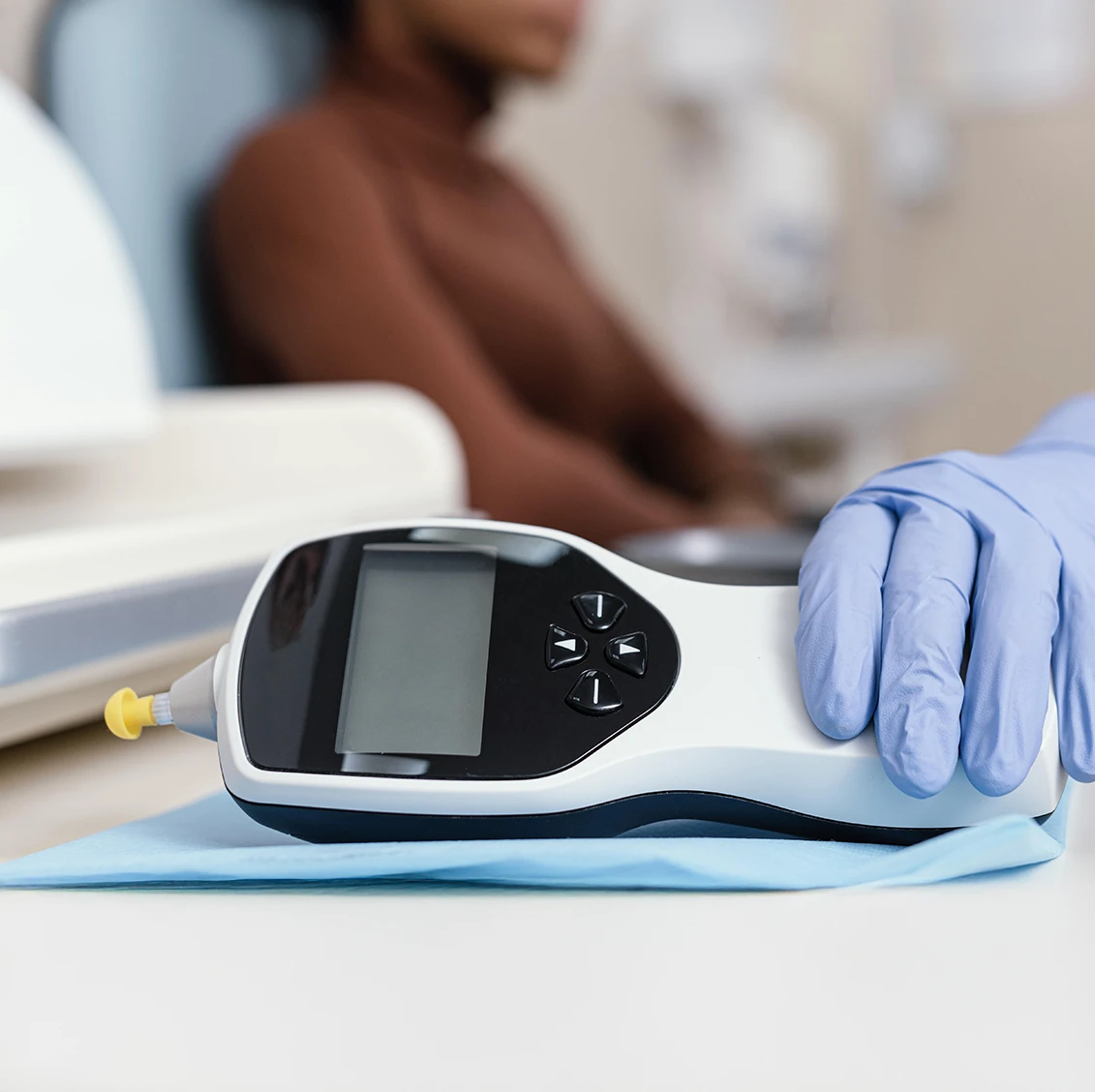
We have supported pricing decisions across a wide range of medtech solutions, including tele-consultation services, ultrasound systems, vital signs monitoring, diabetes testing devices, and endoscopic technologies.
How We Have Helped Med-tech Firms With Pricing Research
Our Pricing Research Toolkit
We apply rigorously tested quantitative techniques, tailored to medtech use cases, including

Conjoint analysis to model feature-level value trade-offs

Gabor-Granger to assess price elasticity and demand curves

Van Westendorp pricing models to define acceptable price ranges
From Analysis to Pricing Strategy
We translate data into clear pricing guidance that informs portfolio architecture, market entry strategy, and revenue optimization. By anchoring price to perceived clinical and economic value, EMeRG helps medtech companies price with confidence and compete effectively.
Conference Research
Independent Technology Intelligence for Hospital Decision-Makers
Hospitals increasingly rely on industry conferences to stay ahead of medical technology innovation. At EMeRG, we use global and regional medtech conferences as structured intelligence-gathering platforms to support informed procurement and technology adoption decisions.
We act as an independent extension of healthcare systems, attending major medtech conferences to evaluate emerging technologies from both clinical and operational perspectives. Our work begins well before the event with focused analysis of exhibitors, speakers, and innovation themes. This is followed by structured on-ground engagements with technology providers and key stakeholders.
Led by experienced healthcare analysts and grounded in ethnographic observation, each interaction delivers unbiased insight into technology maturity, differentiation, and real-world applicability. All conference research is conducted under strict ethical standards. Through this approach, EMeRG helps hospitals cut through noise, reduce uncertainty, and make confident, evidence-based technology decisions.


Value Proposition Research
Understanding the deeper value drivers and motivators of a customer segment is critical in designing value proposition statements that would find instant resonance with the target segments. At EMeRG, we look not only into the “stated importance” but also the “derived importance” of benefits sought by customers. Our granular approach including various qualitative and quantitative methodologies are aimed at ensuring that a distinct Rivalry proposition is created for our med-tech clients. EMeRG has helped med-tech firms evaluate, test and refine value proposition statements for Ultrasound, tele-health solutions, diabetes testing devices, and patient monitoring devices (among others).